CNRS opinion on the works of Jacques Benveniste
Six months after his death, the CNRS opinion on the works of J. Benveniste
March 8, 2005
Preamble:



**
[The audio file of the interview with Montagnier, May 2010](../../AUDIOS/LE SEPT NEUF DU DIMANCHE 02.05.2010_benveniste.mp3)
May 10, 2010.
A reader sent me excerpts from a program where you can hear the Nobel Prize winner Luc Montagnier praising my late friend Jacques Benveniste.
Professor Luc Montagnier, Nobel Prize in Medicine, in 2007, at Lugano, he does not hold back and does not hesitate to declare that Jacques was a brilliant pioneer, ahead of his time, and his conviction that one day the correctness of his views would be recognized.
I remember the time when the general director of the INSERM, Lazare, removed from Jacques his 200 square meters of premises at the INSERM 200 in Clamart, which led him to move back to the Algeco barracks in the courtyard! A complete disgrace.
Many times I told Jacques, "give up, you will lose your life!" But he held on, held on, until his last breath, until he lost his life, with a broken heart.
My career has had similar aspects and I owe my life to the fact that it has been nothing but an uninterrupted series of abandonments: MHD in 1972 (by abandoning at the Institute of Fluid Mechanics in Marseille the installation with which I had brought the lab to the forefront internationally in 1967), computer science in 1983 (I was deputy director of the computer service of the University of Provence), teaching in the faculty of letters, mathematics (the sphere inversion, Pour la Science 1979), a comeback in MHD (1975-1986), abandonment of comic book publishing with a publisher, in 1990, a quick abandonment, in the years 2000, in Egyptology. Currently, almost abandonment, or serious inactivity in astrophysics, cosmology and mathematical physics, due to lack of positive responses (1985-2008).
Currently, a rebound with Savoir sans Frontières and the reissue of books and comics. Activities on the edge of abandonment in MHD and UFO topics. Below is the photo of the MHD bench in the process of assembly in Rochefort (status in May 2010):
It is in the style of the Algeco of Jacques, in the courtyard of the INSERM, the difference being that it is not me who is in charge, but a courageous 40-year-old technician. Unlike Bernard Palissy, I will not burn my furniture.
The French leading edge MHD, the "out of equilibrium" MHD, the "bitemperature plasmas", which allows us to be among the top in international conferences (Vilnius 2008, Bremen 2009), here it is!
It would be funny if it weren't so utterly sad
On the occasion of the year of physics, the magazine Le Courrier du CNRS published a brochure presenting ten unresolved problems in science. One of these questions concerns the structure of water, in all its states. Below is the reproduction of this dossier.

 | For now, physicists have managed to observe that they are constantly created and destroyed - each lives on average a thousandth of a billionth of a second, that a large number of them are formed and that the three atoms involved must be perfectly aligned for a bond to appear. Now, of all the liquids, water is the only one to have these three characteristics. And this is probably what explains in part the famous anomalies described by researchers: on one hand, water is not a gas at room temperature, because the hydrogen bonds are strong enough. Water therefore has a great cohesive force. Result: a lot of energy must be brought in to break these bonds, which explains why it only boils at 100 °C. On the other hand, it is not a solid at room temperature, because the bonds are still fragile. |
| For now, physicists have managed to observe that they are constantly created and destroyed - each lives on average a thousandth of a billionth of a second, that a large number of them are formed and that the three atoms involved must be perfectly aligned for a bond to appear. Now, of all the liquids, water is the only one to have these three characteristics. And this is probably what explains in part the famous anomalies described by researchers: on one hand, water is not a gas at room temperature, because the hydrogen bonds are strong enough. Water therefore has a great cohesive force. Result: a lot of energy must be brought in to break these bonds, which explains why it only boils at 100 °C. On the other hand, it is not a solid at room temperature, because the bonds are still fragile. |
|---|
** | P | our
| P | our
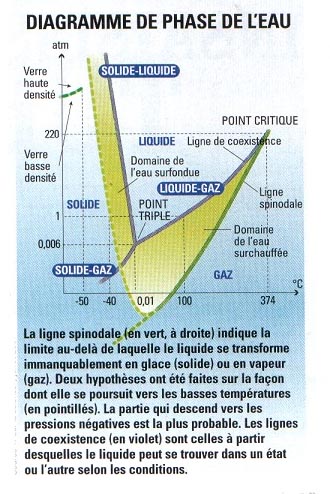
| José Teixeira, a deeper understanding of the dynamics of the hydrogen bond is essential if we want to really understand water one day. Bernard Cabane also goes in this direction: "We still lack a lot of information about water to have a realistic and predictive model of its behavior. Even though we know the nature of the bonds between molecules, as long as we don't know how an isolated molecule interacts not only with its first neighbors but also with others, the model will not be good. And current numerical simulations agree with this. Indeed, if we try to account for the three main peculiarities of water, the models only reproduce one or two. Never all three at the same time. Hydrogen bond: it is established between two identical or non-identical molecules. It is the lack of charge carried by the hydrogen atom that allows the bond to appear. But physicists are not short of ideas to try to solve the mystery. For this, they study its structure at low temperature. "Hydrogen bonds are more stable below 0 °C," explains José Teixeira. "We can then try to better understand liquid water if we follow its evolution up to - 40°C." Liquid up to -40 °C? Yes, if it is free of all impurities, otherwise it crystallizes immediately. Scientists call this supercooling (see phase diagram), which also exists for other liquids, such as toluene, gallium or molten silica. "For now, the record for water is at - 42 °C - barely better than for supercooled water present in certain atmospheric clouds," says Frédéric Caupin, researcher at the Laboratoire de physique statistique de l'ENS. Below - 40 °C, nothing but the thermal agitation of water molecules seems sufficient for the liquid to turn into ice. Once this temperature barrier is passed, the lifetime of liquid water becomes extremely brief. Physicists then no longer have the means to observe it. |
|---|
It is around -130 °C that another interesting phenomenon appears: if water is cooled sufficiently to this temperature, it turns into amorphous ice, that is, it has the structure of glass (see p. 16). A fact becomes clear: "We know nothing about the structure of water between - 40 °C and -130 °C," admits José Teixeira. With a bit of humor, researchers call this area the "no man's land." A twist in 1984: physicists Mishima, Calvert and Whalley discovered a second form of amorphous ice, denser than the first, by compressing ordinary ice at very low temperatures. A result that has revived old ideas. Indeed, in 1892, Röntgen hypothesized that water was a mixture of liquid and ice. Today, some see in the discovery of the two forms of amorphous ice a promising lead: water would be, at least at low temperatures, a mixture of two liquids, one of low density and one of high density. An idea that leaves José Teixeira a bit skeptical. And he suggests that the culprit is still and always the hydrogen bond. But how to decide, when the no man's land remains inaccessible to measurements? A solution: improve the so-called cavitation experiments, which take place at room temperature and at pressures called "negative."
For José Teixeira, a deeper understanding of the dynamics of the hydrogen bond is essential if we want to really understand water one day. Bernard Cabane also goes in this direction: "We still lack a lot of information about water to have a realistic and predictive model of its behavior. Even though we know the nature of the bonds between molecules, as long as we don't know how an isolated molecule interacts not only with its first neighbors but also with others, the model will not be good. And current numerical simulations agree with this. Indeed, if we try to account for the three main peculiarities of water, the models only reproduce one or two. Never all three at the same time. Hydrogen bond: it is established between two identical or non-identical molecules. It is the lack of charge carried by the hydrogen atom that allows the bond to appear. But physicists are not short of ideas to try to solve the mystery. For this, they study its structure at low temperature. "Hydrogen bonds are more stable below 0 °C," explains José Teixeira. "We can then try to better understand liquid water if we follow its evolution up to - 40°C." Liquid up to -40 °C? Yes, if it is free of all impurities, otherwise it crystallizes immediately. Scientists call this supercooling (see phase diagram), which also exists for other liquids, such as toluene, gallium or molten silica. "For now, the record for water is at - 42 °C - barely better than for supercooled water present in certain atmospheric clouds," says Frédéric Caupin, researcher at the Laboratoire de physique statistique de l'ENS. Below - 40 °C, nothing but the thermal agitation of water molecules seems sufficient for the liquid to turn into ice. Once this temperature barrier is passed, the lifetime of liquid water becomes extremely brief. Physicists then no longer have the means to observe it.
For José Teixeira, a deeper understanding of the dynamics of the hydrogen bond is essential if we want to really understand water one day. Bernard Cabane also goes in this direction: "We still lack a lot of information about water to have a realistic and predictive model of its behavior. Even though we know the nature of the bonds between molecules, as long as we don't know how an isolated molecule interacts not only with its first neighbors but also with others, the model will not be good. And current numerical simulations agree with this. Indeed, if we try to account for the three main peculiarities of water, the models only reproduce one or two. Never all three at the same time. Hydrogen bond: it is established between two identical or non-identical molecules. It is the lack of charge carried by the hydrogen atom that allows the bond to appear. But physicists are not short of ideas to try to solve the mystery. For this, they study its structure at low temperature. "Hydrogen bonds are more stable below 0 °C," explains José Teixeira. "We can then try to better understand liquid water if we follow its evolution up to - 40°C." Liquid up to -40 °C? Yes, if it is free of all impurities, otherwise it crystallizes immediately. Scientists call this supercooling (see phase diagram), which also exists for other liquids, such as toluene, gallium or molten silica. "For now, the record for water is at - 42 °C - barely better than for supercooled water present in certain atmospheric clouds," says Frédéric Caupin, researcher at the Laboratoire de physique statistique de l'ENS. Below - 40 °C, nothing but the thermal agitation of water molecules seems sufficient for the liquid to turn into ice. Once this temperature barrier is passed, the lifetime of liquid water becomes extremely brief. Physicists then no longer have the means to observe it.

| E | lles
| test the cohesion of water by searching for the maximum traction that can be exerted with ultrasound on liquid water before the first vapor bubble forms. "The hope is to reach a pressure of -1400 bars," comments Frédéric Caupin. "Then, we would have new elements that would allow us to rule out certain hypotheses on the structure of water." Unfortunately, these experiments are very difficult to perform today. Progress still remains to be made in the purification of water. Patience, therefore. The mystery of water is likely to puzzle researchers for many years to come, says Julien Bourdet. To solve the mysteries of water, physicists rely on cavitation experiments (here carried out in a hydrodynamic tunnel) where water vapor bubbles appear. |
|---|
Among the anomalies of water, the three main ones are
-
a very strong cohesion, which is manifested by high melting and boiling points;
-
a high dielectric constant, which allows it to dissolve all salts.
To solve the mystery of water, physicists rely on cavitation experiments (here carried out in a hydrodynamic tunnel) where water vapor bubbles appear
- a large expansion at low temperature (below 4 °C) and also during crystallization.
Just as supercooled water can be found, so can superheated water, that is, liquid above 100 °C. The explosive appearance of a bubble is called cavitation. A drop in pressure is equivalent to heating the water. Researchers stretch the water (they speak of negative pressure) until they observe the first vapor bubble.
**Julien Bourdet **
CONTACTS
Bernard Cabane : bcabane @ pmmh.espci.fr Frédéric Caupin : caupin @ lps.ens.fr José Teixeira : teix@ Ilb.saclay.ceafr

Among the anomalies of water, the three main ones are
-
a very strong cohesion, which is manifested by high melting and boiling points;
-
a high dielectric constant, which allows it to dissolve all salts.
To solve the mystery of water, physicists rely on cavitation experiments (here carried out in a hydrodynamic tunnel) where water vapor bubbles appear
Among the anomalies of water, the three main ones are
-
a very strong cohesion, which is manifested by high melting and boiling points;
-
a high dielectric constant, which allows it to dissolve all salts.
To solve the mystery of water, physicists rely on cavitation experiments (here carried out in a hydrodynamic tunnel) where water vapor bubbles appear
Vers " Opinion du Cnrs sur les travaux de Jean-Pierre Petit "
Retour vers Guide Retour vers page d'Accueil
Number of visits to this page since March 8, 2005 :